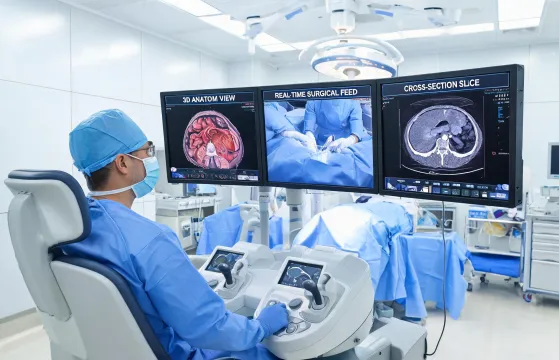
Console-guided procedure planning
Procedure setup cards define instrument families, OR turnover signals, training checkpoints, and case-support escalation paths before the room is live.
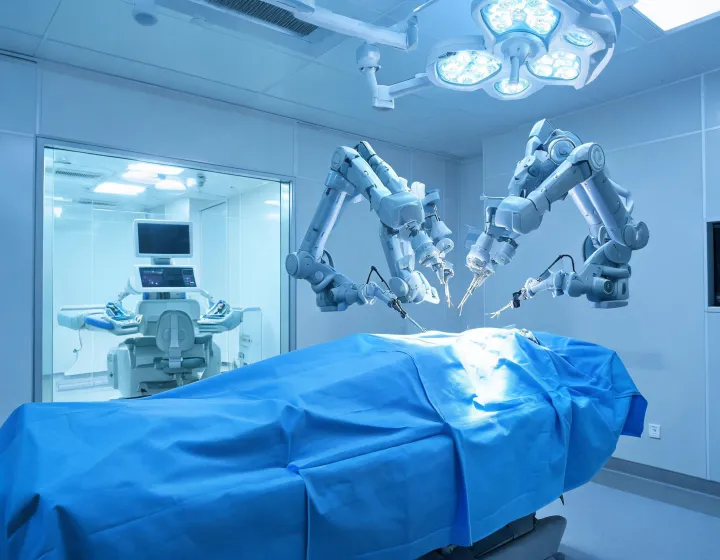
Intuitive Surgical brings robotic instrumentation, remote care signals, and clinical workflow data into a disciplined operating model for hospitals expanding minimally invasive programs.

Procedure setup cards define instrument families, OR turnover signals, training checkpoints, and case-support escalation paths before the room is live.

Observation push, role-based access, SBOM review, and privacy controls help clinical engineers evaluate connected care without losing audit traceability.

Laboratory teams can align LIS middleware, calibration intervals, reagent lot traceability, and result routing with perioperative data review.
Share the care setting, target procedures, integration constraints, and service expectations. A specialist will map the evaluation pathway.
Start Evaluation